On the surface, the flexible endoscopy industry might seem to be business as usual: gastroscopes, colonoscopes, bronchoscopes, biopsy forceps, hemostatic clips, snares, and disposable cholangoscopes…
However, a comparative analysis of the 2025 annual reports and Q1 2026 interim reports of Micro-Tech and Sonoscape reveals a distinct trend:
China’s flexible endoscopy sector has moved beyond the narrative of “import substitution.” Instead, it has entered a new phase of comprehensive competition encompassing equipment, consumables, technology, clinical application, training, after-sales service, and the broader ecosystem.
Editor‘s Note:
As competition in the flexible endoscopy industry shifts from “product substitution” to a “full-stack clinical ecosystem,” the role of endoscopic accessories is evolving. They are no longer just supporting tools but essential components of the therapeutic platform.
ZRHmed, a manufacturer of endoscopic accessories, supplies MDR CE certified products including hemostatic clips, polypectomy snares, injection needles, biopsy forceps supporting both GI and Endourology procedures with reliable quality.
Micro-Tech represents the direction of endoscopic consumables and disposable endoscopes.
Sonoscape embodies the trajectory of domestic flexible endoscopy equipment and smart endoscopy platforms.
One solves “how to treat”;
The other addresses “how to visualize”.
One goes deep into the treatment side;
The other builds strength on the equipment side.
These two companies are, in essence, the two best windows through which to observe China’s flexible endoscopy industry.

I. Let’s Look at the Data: Growth Confirmed for 2025, Pressure Emerging in Q1 2026
When analyzing the industry, we can’t just talk about trends; we must look at the financial reports first.
1.Micro-Tech: Revenue Continues to Grow, Profits Under Short-term Pressure
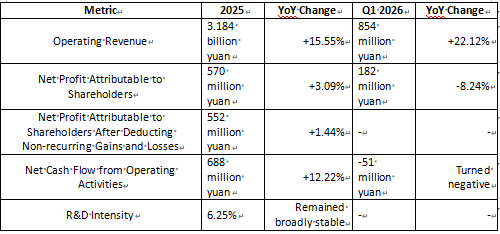
For the full year 2025, Micro-Tech (Nanowei Medical) achieved operating revenue of approximately 3.184 billion yuan, a year-on-year increase of 15.55%. Net profit attributable to shareholders was approximately 570 million yuan, up 3.09% year-on-year. Net profit attributable to shareholders after deducting non-recurring gains and losses was approximately 552 million yuan, up 1.44% year-on-year. Net cash flow from operating activities reached approximately 688 million yuan, a year-on-year increase of 12.22%.
However, in the first quarter of 2026, while Micro-Tech achieved operating revenue of approximately 854 million yuan (up 22.12% year-on-year), the net profit attributable to shareholders was approximately 182 million yuan, representing a 8.24% decline year-on-year. Notably, the net cash flow from operating activities was approximately -51 million yuan.
Micro-Tech Key Financial Data
What Do These Figures Tell Us?
First, Micro-Tech’s revenue growth is solid. In particular, the revenue growth rate in Q1 2026 reached 22.12%, which is even faster than the full-year growth rate of 2025.
Second, profits did not grow in sync with revenue; instead, net profit attributable to shareholders actually declined in Q1 2026. This indicates that the company is facing pressure in areas such as sales, R&D, overseas expansion, exchange rate fluctuations, and product mix adjustments.
Third, operating cash flow was very strong in 2025, reaching 688 million yuan—higher than the net profit attributable to shareholders—suggesting that Micro-Tech’s overall operational quality is solid. However, the fact that operating cash flow turned negative in Q1 2026 serves as a reminder: during this expansion phase, we need to keep a close eye on inventory stocking, accounts receivable, and expense investments.
To sum up in one sentence: Micro-Tech’s business is still growing, but it has transitioned from a “period of high-growth dividends” to a “period of growth quality testing.”
2.Sonoscape Medical: Revenue Growth Resumes, Profit Volatility Becomes More Pronounced
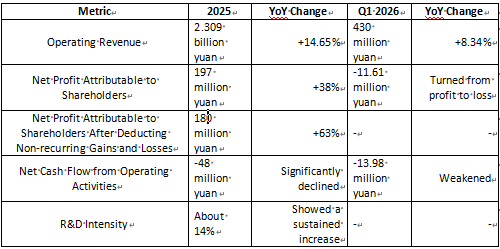
In 2025, Sonoscape Medical achieved operating revenue of approximately 2.309 billion yuan (23.09 hundred million), a year-over-year increase of 14.65%. Net profit attributable to shareholders was about 197 million yuan, up roughly 38%, while net profit attributable to shareholders after deducting non-recurring gains and losses reached 180 million yuan, surging by approximately 63%.
However, net cash flow from operating activities was approximately -48 million yuan, marking a significant decline compared to 2024.
In the first quarter of 2026, Sonoscape Medical recorded operating revenue of about 430 million yuan, a year-over-year growth of 8.34%. Net profit attributable to shareholders was approximately -11.61 million yuan, turning from profit to loss. Net cash flow from operating activities stood at about -13.98 million yuan.
Sonoscape Medical Key Financial Data
Sonoscape Medical: Two Key Characteristics
First, both revenue and profit resumed growth in 2025. This indicates that the recovery in medical equipment tendering and procurement, growth in endoscopy business, and the promotion of high-end products have provided solid support for the company’s performance.
Second, the loss in Q1 2026 highlights that equipment manufacturers face more pronounced quarterly fluctuations. The medical device industry involves complex cycles of bidding, installation, acceptance, and payment collection, so quarterly profits cannot be simply extrapolated linearly.
This is especially true for products like flexible endoscopy systems. It’s not a case of “sell today, recognize all value tomorrow.” Behind every sale lies a comprehensive process involving installation, training, clinical onboarding, after-sales service, and cultivating experts’ usage habits.
To sum up in one sentence: Sonoscape Medical has established itself as the domestic leader in flexible endoscopy equipment, but breaking through in the high-end market requires continuous investment, making short-term profit fluctuations a normal phenomenon.
II. Putting Both Companies Together, the Industry Logic Becomes Clear
Micro-Tech and Sonoscape are not the same type of company. Micro-Tech is more of a platform enterprise for endoscopic therapeutic consumables, while Sonoscape resembles a platform enterprise for domestic flexible endoscopy equipment.
However, they both point to a single trend: the future of the flexible endoscopy industry will not be about competition between single products, but competition based on holistic solutions centered around the endoscopy center.
Micro-Tech Medical: The Core Logic of Endoscopic Therapeutics
Micro-Tech’s primary product portfolio consists of three major categories:
Endoscopic Diagnosis and Treatment Devices: Including biopsy, hemostasis and closure, EMR/ESD, dilation, ERCP, and EUS/EBUS devices.
Microwave/RF Ablation Equipment and Consumables.
Disposable Endoscopes: Including disposable biliary-pancreatic imaging systems, disposable cholangioscopes, and disposable bronchoscopes.
In other words, Micro-Tech is not simply selling consumables; it is building a product matrix centered around endoscopic therapeutic scenarios.
For instance, the deeper a digestive endoscopy center delves into procedures, the greater its demand for Micro-Tech’s products: biopsy forceps for biopsies, snares for polypectomy, hemostatic clips for bleeding, ESD devices for early cancer treatment, ERCP consumables for biliary-pancreatic diseases, needles for EUS/EBUS, and disposable cholangioscopes for direct visualization of the bile duct.
Therefore, Micro-Tech’s growth essentially stems from two directions:
1.An increase in the volume of endoscopic examinations.
2.An increase in the penetration rate of endoscopic therapeutics.
The second point is what truly matters. This is because examinations drive the consumption of basic consumables, whereas therapeutics drive the demand for high-value and complex consumables.
Sonoscape Medical: The Core Logic of High-End Domestic Flexible Endoscopes
Sonoscape Medical represents the breakthrough on the equipment side.
In its 2025 annual report, Sonoscape highlighted that it was one of the earliest domestic enterprises to develop and master core technologies in medical endoscopic equipment and scopes. It ranks third in the domestic digestive endoscopy market, trailing only Olympus and Fujifilm, with its domestic market share rising to approximately 14%.
This data is critical. Historically, the domestic flexible endoscope market has been dominated by imported brands—particularly Olympus, Fujifilm, and Pentax—which enjoy strong usage habits and brand trust among physicians.
For domestic brands to break through, low pricing alone is not enough. What physicians truly care about includes:
Image clarity;
Smoothness of insertion;
Ease of scope manipulation;
Stability of the water/air supply system;
Usability of the biopsy channel;
Durability of the scope;
Timeliness of after-sales response;
Capability to support complex therapeutic scenarios.
The significance of Sonoscape lies in the fact that it is not merely pursuing low-end substitution. Instead, it is advancing towards high-end flexible endoscopes, specialized scope types, AI quality control, 4K platforms, and multi-scope combined solutions.
III. The First Major Shift in China’s Flexible Endoscopy Industry: From “Examination Tool” to “Therapeutic Platform”
In the past, many hospitals had a simple understanding of flexible endoscopes: gastroscopes and colonoscopes were primarily for examination. But today, flexible endoscopy is no longer just an examination tool; it has evolved into a diagnostic and therapeutic platform.
It is now capable of performing:
Digestive tract early cancer screening;
Biopsy sampling;
Polypectomy;
EMR (Endoscopic Mucosal Resection);
ESD (Endoscopic Submucosal Dissection);
ERCP (Endoscopic Retrograde Cholangiopancreatography);
EUS (Endoscopic Ultrasound) puncture;
EBUS (Endobronchial Ultrasound) puncture;
Direct visualization and treatment of the bile duct;
Bronchoscopy diagnosis and treatment;
Hemostasis;
Closure;
Dilation;
Stent placement;
Local ablation.
Therefore, the fundamental change in the industry is a shift from simply “taking a look” to being able to “see clearly, access, resect, stop bleeding, and treat effectively.”
This is precisely why both endoscopic consumable companies and endoscopic equipment companies are upgrading:
On the equipment side: To achieve clearer visualization.
On the consumables side: To enable more precise treatment.
On the training side: To empower physicians to perform more complex techniques.
On the after-sales side: To ensure the continuous operation of endoscopy centers.
IV. Market Dynamics: Domestic Substitution Enters Deep Waters
The Chinese flexible endoscopy market has long been dominated by imported brands. However, domestic substitution has now entered its second phase.
Phase 1: Price-based Substitution
In the early stage, domestic flexible endoscopes primarily entered hospitals by leveraging a price advantage. Imported brands were expensive; domestic brands were cheaper.
Some primary care hospitals, secondary hospitals, and institutions with limited budgets were willing to try domestic products. However, substitution at this stage was relatively superficial. Physicians might have treated domestic scopes merely as backup units, not as their primary devices.
Phase 2: Performance-based Substitution
The industry is now entering the stage of performance-based substitution. This means domestic products must truly approach, or even partially surpass, imported brands in clinical performance.
Sonoscape Medical’s 4K iEndo Smart Endoscopy Platform HD-650 series, launched in 2025, is a prime representative of this direction. According to Sonoscape’s annual report, the company has achieved real-time super-resolution imaging for 4K endoscopic video in imaging technology and digital image processing. It has also mastered technologies such as fusion spectral imaging, spectrum aggregation imaging, photoelectric composite chromoendoscopy, multispectral imaging, low-latency image processing, and real-time 4K ultra-high-definition digital image processing.
These technologies are not merely for impressive specifications; they directly serve clinical needs:
Clearer visualization of early cancer boundaries;
Finer mucosal texture;
Easier identification of microvascular structures;
More distinct color differences in lesions;
Lower image latency;
Greater stability for physicians during prolonged procedures.
This is exactly where the key lies for whether domestic flexible endoscopes can enter the core scenarios of Grade IIIA hospitals.
Phase 3: Ecosystem-based Substitution
Going a step further, future domestic substitution will not be about simply replacing a single scope or a single consumable. Instead, it will be about replacing the “usage ecosystem” long established by imported brands.
This ecosystem includes:
Equipment;
Scopes;
Imaging systems;
Specialized scope types;
Therapeutic consumables;
Physician training;
Nurse training;
After-sales maintenance;
Academic promotion;
Clinical pathways;
Quality control systems.
In the future, hospitals will not just be buying products, but complete capabilities.
Therefore, the competition in the flexible endoscopy industry is shifting from “product substitution” to “ecosystem substitution.”
About ZRHmed
ZRHmed is a Chinese manufacturer specializing in endoscopic accessories. Our product range includes hemostatic clips, polypectomy snares, injection needles, biopsy forceps, cleaning brushes, and ureteral access sheaths. Core products are MDR CE certified and exported to Europe, the Middle East, Southeast Asia, and South America. We are committed to being a trusted partner for endoscopists worldwide.
Post time: May-13-2026


