Latest industry research data shows: In 2025, China’s endoscope export value reached RMB 4.47 billion, a year-on-year increase of 32.48%. Five years ago, this figure was less than RMB 1 billion — representing a compound annual growth rate (CAGR) of 35.14%. Endoscopy, a field once firmly dominated by international giants such as Olympus and Stryker, is rapidly becoming a “star product” for China’s medical device industry going global.
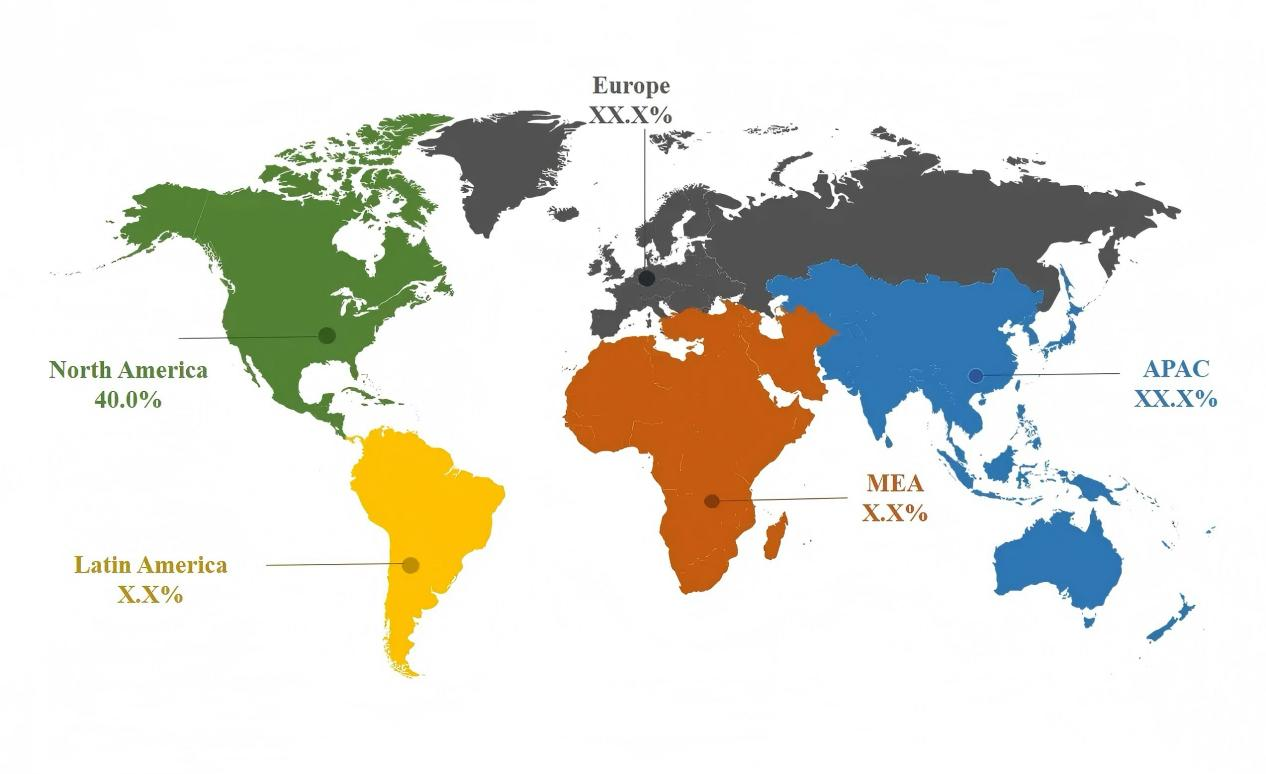
The top five export destinations are the United States, Germany, France, Russia, and Brazil, with exports to the U.S. market alone exceeding RMB 1 billion.
So, which Chinese companies are leading this charge, and what internationalization paths have they taken? This article provides a comprehensive overview.
1. Global Strategies of Comprehensive Industry Leaders
Mindray Medical
As the undisputed leader in China’s medical device industry, Mindray’s endoscope products have long reached global markets through its extensive overseas distribution channels. In the first half of 2025, Mindray’s overseas business revenue reached RMB 8.332 billion, accounting for 50% of total revenue for the first time. In Q3, international business grew 11.93% year-on-year, with the European market growing over 20%. Mindray’s management has clearly stated its goal to gradually increase the overseas revenue share to 70%. For endoscopy, Mindray’s brand equity and channel synergy represent a moat difficult for others to replicate.
Shinva Medical
Shinva Medical has entered the endoscopy field through its subsidiary, Shinva Surgical Instruments Co., Ltd. In 2025, its self-developed medical endoscope camera system obtained EU CE certification, including 4 MDR certificates covering 7 products. Orders have come from multiple countries, including Bangladesh, Chile, Ukraine, and Iraq — while individual order sizes are modest, the geographic coverage is broad, indicating strong demand from emerging markets.
2. How Are Single-Use Endoscope Companies Performing?
If the endoscope export drive is an army, then single-use endoscope companies are the tip of the spear. In 2025, the following companies delivered particularly outstanding performances.
HugeMed Medical:
80% Annual Growth, 103 Global Certifications
Founded in 2014, HugeMed is a leader in Chinese single-use endoscope exports and the first Chinese single-use endoscope company to receive CE MDR certification. To date, the company has obtained 103 medical product certifications across major global markets, including NMPA, CE, and FDA.
Its product pipeline covers six major clinical scenarios: anesthesiology/emergency/ICU, pulmonology, urology, etc., with an annual production capacity of 400,000 endoscopes. In 2025, HugeMed’s revenue grew by over 80% and secured a RMB 100 million+ financing round co-led by Fengnian Capital and Qiming Venture Partners.
Vathin Medical:
Deeply Tied to Olympus, Acquisition of MGB, a century-old German rigid endoscope company
Vathin Medical is a typical example of “going global first, then domestic.” Through deep collaboration with international giants like Olympus, Medi-Globe, and TKB, and acquiring the century-old German rigid endoscope company MGB, Vathin has achieved a full industry chain layout integrating flexible and rigid endoscopy.
Its self-developed single-use endoscopes have entered high-end markets in Europe and the U.S., achieving leading market share in dozens of countries and shipping over 500,000 units cumulatively. The company has obtained over 210 global registrations and certifications and ranks first in single-use bronchoscope market share in multiple countries.
Micro-Tech (Nanjing):
Overseas Revenue 58%, Cross-Border Acquisition of CME
Micro-Tech is a leader in China’s endoscopic minimally invasive device sector, focusing on three major product lines: digestive endoscopy, tumor intervention, and minimally invasive neurosurgery, covering over 90 countries and regions. In H1 2025, its overseas revenue share increased to 58%, with overseas business revenue growing 45% year-on-year.
Notably, in early 2025, Micro-Tech completed the cross-border acquisition of CME, a European medical device distributor, further expanding its European sales channels — moving from product export to channel export.
Pusen Medical:
Topped China’s Endoscope Export Rankings:
Headquartered in Zhuhai, Pusen Medical once ranked first in China’s endoscope export enterprises. Its products are sold in over 100 countries and regions, serving more than 2,000 hospitals globally and collaborating with world-renowned institutions such as the Mayo Clinic and Cleveland Clinic.
In the sub-segment, Pusen’s single-use urological endoscope exports rank first in China, while its overall endoscope exports rank second.
Redpine Medical:
Joint R&D with Cook Medical
Redpine Medica has established deep collaboration with Cook Medical, a global leader in urology. In 2024, Cook promoted Redpine’s single-use ureteroscope products in the North American market; in 2025, the two companies further signed a multi-million dollar joint R&D agreement to develop a single-use large-channel pyeloscope with real-time pressure measurement capabilities.
MacroLux Medical:
Secured Global Distribution Agreement with Olympus
Founded in 2020, MacroLux Medical’s founding team comes from leading domestic and international device companies. In 2025, it reached a global distribution agreement with Olympus, granting Olympus exclusive distribution rights for its single-use urology products in the U.S., Canada, and parts of Europe and Asia-Pacific — a typical case of a Chinese startup leveraging an international giant’s channel.
Other Emerging Players Not to Be Overlooked
Innovex Medical: Based in Shanghai Zhangjiang, a provider of non-vascular interventional total solutions. Single-use endoscope overseas revenue accounts for over 80% of total overseas revenue. Products feature a world-first 800×800 pixel CMOS high-definition imaging and have received certifications in China, the U.S., Europe, and Japan.
Endoso Life: Revenue grew over 80% in 2025, tied with HugeMed Medical as the fastest-growing single-use endoscope company. Products cover urology, pulmonology, gynecology, and other departments, with rapid overseas market expansion.
HAPPINESS WORKS MEDICAL: Focused on single-use ultra-fine electronic endoscopes. In 2025, single-use urology product sales volume grew 35% year-on-year. Products include ureteroscopes, cystoscopes, bronchoscopes, etc., with continued overseas efforts.
Seegen Optoelectronics:Has achieved scaled sales and stable profit in single-use endoscopy, offering differentiated products in urology and other fields, with steady overseas market expansion and significant growth potential.
- Progress in Flexible Endoscopy and Capsule Endoscopy
Aohua Endoscopy:
Acquired German WISAP, Overseas Sales Up 42.7%
Aohua Endoscopy is a representative Chinese company in electronic endoscopy. In 2018, it acquired Germany-based WISAP, gaining a local production, marketing, and after-sales team. Its sales are primarily in Europe and the Rest of the World (ROW), with high-end model coverage established from scratch in markets such as Brazil, South Korea, Malaysia, Thailand, and Myanmar.
In 2024, Aohua’s overseas sales grew 42.7% year-on-year, far exceeding its domestic growth rate. Its next-generation flagship model, the AQ400, has been successfully launched, further contributing to revenue growth.
SonoScape Medical:
Dual-Drive of Ultrasound + Endoscopy, Launched Bonded Maintenance in Hainan
SonoScape’s ultrasound diagnostic equipment ranks among the top ten globally in market share, and its digestive and respiratory endoscopes are among the world’s leading products. Its products are sold in over 170 countries and regions. In 2025, SonoScape launched the province’s first medical device bonded maintenance project in the Hainan Free Trade Port, providing standardized maintenance services to global customers — a critical step in servicing globalization.
Ankon Technologies:
World’s Only FDA-Cleared Magnetically Controlled Capsule Gastroscope
Ankon Technologies specializes in magnetically controlled capsule gastroscopy, using an external magnetic field to precisely control capsule movement in the stomach, enabling “non-intubated gastroscopy.” Its greatest moat is being the world’s only capsule gastroscope company with both FDA and EU CE certification. In 2025, Ankon’s small-bowel capsule endoscope entered the U.S. Premier Group purchasing network, and its latest modular intelligent products have been adopted by over 100 healthcare institutions globally, covering the U.S., France, Hungary, Italy, etc., operating at full capacity.
Jinshan Technology:
From Capsules to Surgical Robots — A Product Matrix
Jinshan Technology developed China’s first capsule endoscope, breaking Western monopoly. It has now iterated to the fourth generation, holding over 70% domestic market share, and launched the world’s first fully automated magnetically controlled capsule robot RC100 (CE + NMPA certified). Its products go far beyond capsules: full-HD electronic endoscopes (VE-260), gastrointestinal motility diagnostic systems (MDR certified in 2024), and thoraco-abdominal surgical robots have all gone global, with over 1,300 patents.
Shanghai Huger Medical:
Steady Going Global of an Established Electronic Endoscope Player
Founded in the 1990s, Shanghai Huger Medical is an established Chinese company in medical electronic endoscopy, covering electronic gastroscopes, colonoscopes, bronchoscopes, etc. The company has long pursued a dual-track going global strategy of OEM and own-brand sales, with products exported to Europe, Southeast Asia, South America, and beyond. While not having “viral” products like the former two, it holds a stable position in mid-to-low-end overseas markets thanks to reliable craftsmanship and cost-effectiveness.
- Rigid Endoscopes and Upstream Core Components
NovelBeam:
Deeply tied to Stryker, providing endoscope light source modules and core components, with overseas revenue accounting for over 95%. To mitigate tariff impacts, it has established a plant in Thailand. Next-generation endoscope system is under joint development.
Tiansong Medical:
An established Chinese company in rigid endoscopes, covering arthroscopes, laparoscopes, ENT endoscopes, etc. Long-term focus on overseas OEM and own-brand exports to Europe, the Middle East, and Southeast Asia.
Shenda Endoscope:
A pioneer of rigid endoscopes in China, holding multiple patents and proprietary IP. Products include hysteroscopes, cystoscopes, etc., exported to dozens of countries and regions, including Europe, the U.S., and Southeast Asia.
Hangzhou Optcla Medical:
Representative of the Tonglu medical device cluster, specializing in rigid laparoscopes and supporting minimally invasive surgical instruments. Leveraging high cost-performance advantages, it has long expanded in mid-to-low-end overseas markets with extensive OEM experience.
Hangzhou HAWK:
Focused on urological rigid endoscopes and supporting surgical equipment such as lithotripsy and electrosurgical devices. Products exported to Europe, South America, and the Middle East, with years of OEM export experience and a solid reputation.
- Special Technology Directions:
Endoscopic Ultrasound & Surgical Robots
Inner Medical:
Endoscopic Ultrasound Obtained CE MDR Certification
Inner Medical focuses on three major product lines: endoscopic ultrasound, electronic endoscopes, and vascular imaging systems. Its full range of endoscopic ultrasound and micro-probe products have obtained CE MDR certification. Its world-first integrated mini-probe endoscopic ultrasound has drawn significant attention at international academic conferences.
Harbin Sagebot Intelligent Medical:
Surgical Robot Endoscope Obtained European “Pass”
Harbin Sagebot Intelligent Medical’s Kangduo robotic thoraco-abdominal endoscopic surgical system (SR2000) received EU CE MDR certification in 2025, marking its official qualification to enter the European market.
- Reviewing the Going Global Pathways of Chinese Endoscopes
By examining the internationalization journeys of the above companies, three typical pathways emerge for Chinese endoscope companies going global:
1. Deep Collaboration with Multinational Corporations (MNC)
Vathin Medical partnering with Olympus, Redpine Medical collaborating with Cook Medical, MacroLux Medical granting distribution rights to Olympus. This model enables rapid market access but may limit own-brand development.
2. Overseas Acquisition & Localized Operations
Aohua Endoscopy acquiring Germany’s WISAP, Vathin Medical acquiring Germany’s MGB, Micro-Tech acquiring European distributor CME. Acquisitions are the fastest route to gaining channels, technology, and local teams.
3. Own Brand + Self-Built Channels
HugeMed Medical adhering to an own-brand global strategy; Vathin Medical simultaneously launching own brands and building local teams in Europe and the U.S. This is the most challenging path but offers the highest long-term value.
Why has there been such significant growth among Chinese going-global enterprises in recent years? Domestic market pressure is a major driver — intensifying competition, healthcare cost control, and centralized procurement policies have accelerated the push to go global. However, more fundamental reasons include:
Improved Technological Capabilities: Domestic endoscopes have reached international levels in optical imaging, single-use product manufacturing, and other areas.
Supply Chain Advantages: China’s robust endoscope industry chain is increasingly integrated into global supply chains.
Breakthroughs in Regulatory Approvals: More companies are obtaining international certifications such as FDA and CE MDR, with HugeMed Medical, Vathin Medical, and Enmed Medical among the leaders.
Data Sources: General Administration of Customs of China, Public company financial reports and announcements, Industry research reports.
Thanks for reading. As a manufacturer of endoscopic accessories (biopsy forceps, hemoclip, polyp snare, sclerotherapy needle, spray catheter, cytology brushes, guidewire, stone retrieval basket, nasal biliary drainage cathete etc. which are widely used in EMR, ESD, ERCP. etc.), we are proud to support Chinese endoscope companies going global by providing reliable, CE-marked consumables. If you are an endoscope company looking for a trustworthy accessory partner, let’s connect.
Post time: Apr-15-2026