São Paulo, Brazil – From May 19 to 22, 2026, São Paulo Expo will host one of the most significant milestones in the healthcare sector in Latin America: the 31st edition of Hospitalar. With 33 years of history, the event has established itself as the leading platform for business, innovation, and networking for professionals and companies in the industry.
The 2026 edition promises to be even more special, bringing together leaders, entrepreneurs, and healthcare experts to explore the most advanced solutions, technologies, and trends transforming the market. Get ready for four intense days of strategic connections and unmissable opportunities that will shape the future of healthcare!
Impressive Scale in 2026
According to official information released by the Hospitalar organization, the 2026 edition is expected to welcome approximately 85,000 professional visits, featuring over 1,270 exhibitors from 55 countries, and offering more than 400 hours of content including conferences, seminars, and B2B matchmaking activities. The exhibition will cover an area of over 100,000 square meters, cementing its position as the largest and most comprehensive healthcare trade fair in Latin America.
Extensive Product & Industry Coverage
Hospitalar 2026 spans a wide range of healthcare sectors and product categories, including but not limited to:
Additionally, Hospitalar 2026 will be held concurrently with several specialized co-located shows, including the Brazilian Dental Exhibition (ODONTOBRASIL), Diagnostica (diagnostic technology), Hospfarma (hospital pharmaceuticals), and Reabilitacao (rehabilitation and disability products).
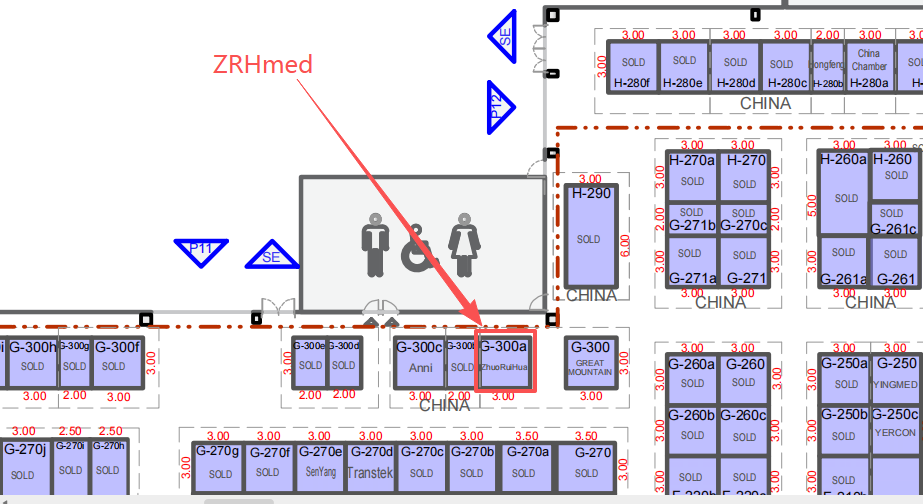
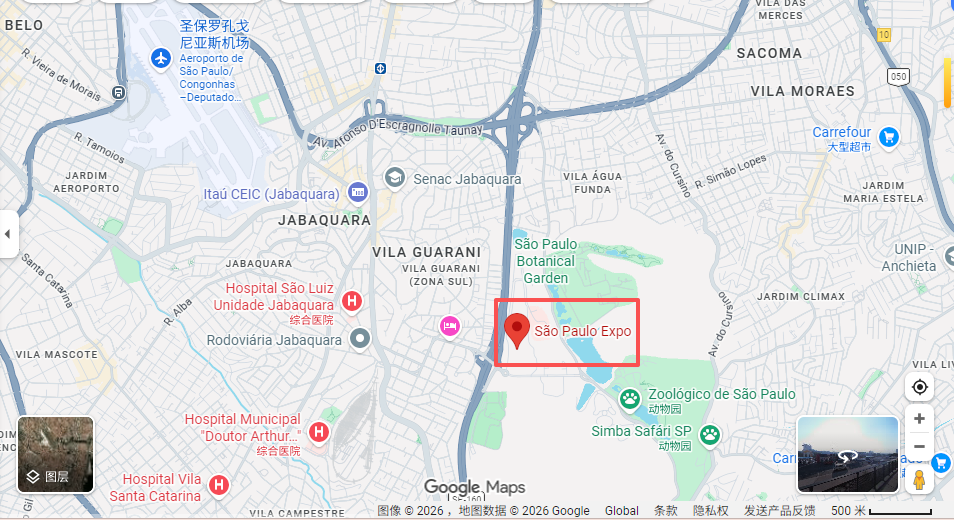
Exhibition Time and Location
Date: 19th to 22nd May, 2026
Time: 11:00 AM to 8:00 PM
Venue: São Paulo Expo | São Paulo – Brasil
About Us
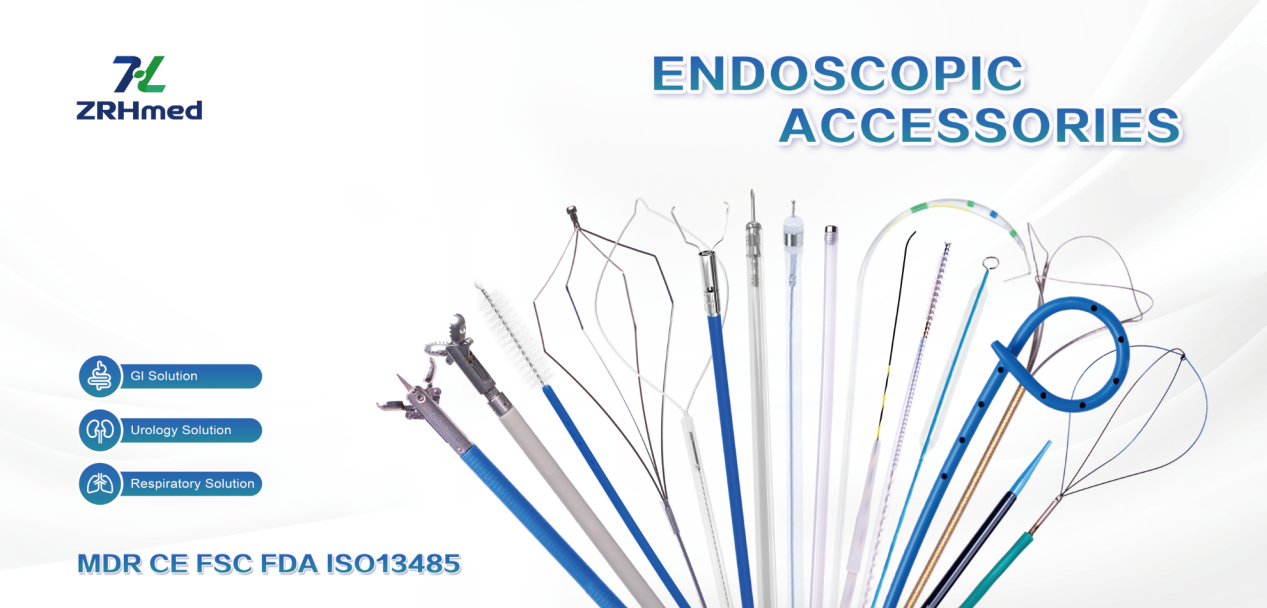
We, Jiangxi Zhuoruihua Medical Instrument Co., Ltd., is a manufacturer in China specializing in the endoscopic consumables, include GI line such as biopsy forceps, hemoclip, polyp snare, sclerotherapy needle, spray catheter, cytology brushes, guidewire, stone retrieval basket, nasal biliary drainage catheter etc. which are widely used in EMR, ESD, ERCP. And Endorology Line, such as ureteral access sheath and ureteral access sheath with suction, disposable Urinary Stone Retrieval Basket, and urology guidewire etc.
Our products are CE certified and with FDA 510K approval, and our plants are ISO certified. Our goods have been exported to Europe, North America, Middle East and part of Asia, and widely obtains the customer of the recognition and praise!
Post time: May-09-2026