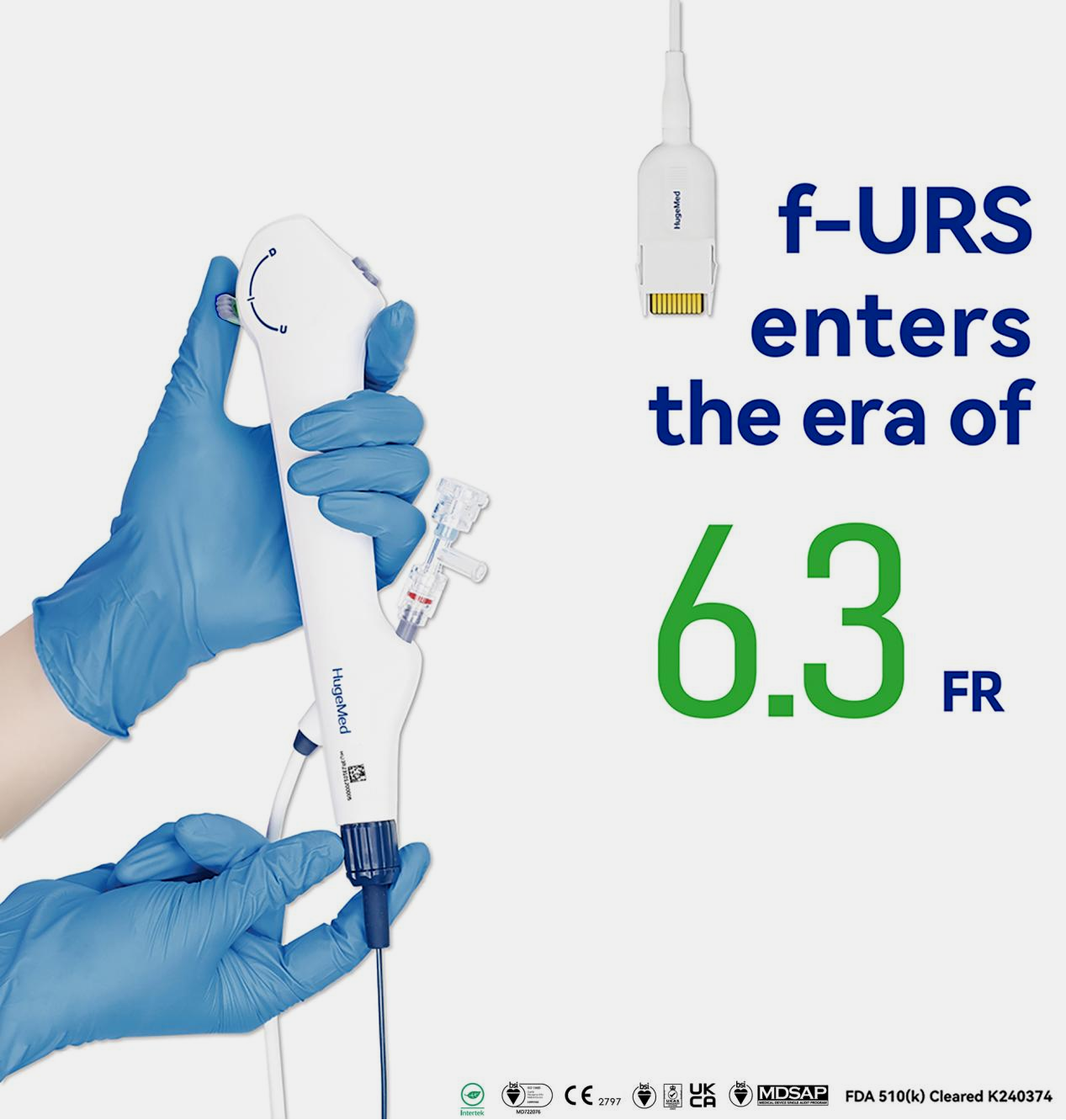
Why Did the 6.3Fr Ultra-Thin Flexible Ureteroscope Become an Industry Benchmark?
On October 10th, 2025, the draft proposal for the sixth round of National Centralized Procurement (NCP) for medical consumables was released. The document set the tone from the outset: "Stabilize clinical practice, ensure quality, prevent bid-rigging, and counter involution."
It is well known that centralized procurement of medical devices concerns both national welfare and people's livelihoods, as well as the development trajectory of related manufacturing enterprises. Therefore, the draft proposal reflects the government's attention to public wellbeing and provides direction for the growth of medical device companies.
Since its inception, HugeMed has adhered to the core principles of "meeting clinical needs and reducing endoscopy costs" as the benchmark for its R&D and supply chain management. Mr. Hu Jifan, Chairman of the company, imbued the enterprise with the vision of "becoming a pivotal force in making endoscopic diagnosis and treatment universally accessible," driven by his initial aspiration of "having a heart for wellness and benefiting the masses.”
"We not only think this way, but we have consistently acted upon it," Chairman Hu Jifan reflected emotionally, recalling the company's ten-year entrepreneurial journey.
HugeMed's vision and values align remarkably well with the philosophy of this round of NCP.
Pioneering the World's First 6.3Fr Single-Use Flexible Ureteroscope Catheter
A Pioneering Leap Guided by Clinical Needs
Flashback to 2022. The global urology market was gradually recovering. HugeMed's international marketing team traveled worldwide with their newly launched 7.5Fr Single-Use Flexible Ureteroscope (URS), extensively gathering clinical feedback. At that time, the 7.5Fr URS was already the industry's slimmest product. However, while praising its quality, experts frequently suggested: "It would be even better if the outer diameter could be reduced further."
When this feedback reached Chairman Hu Jifan, he made an immediate decision, thus launching the R&D project for the 6.3Fr URS.
After two years of intensive R&D investment, HugeMed successfully overcame the technical challenge of reducing the insertion tube's outer diameter to 6.3Fr while maintaining a standard 3.6Fr working channel. This resulted in the world's first 6.3Fr URS with a 3.6Fr channel.
A decisive investment made to fulfill a clinical need has today become the industry benchmark for similar products.
6.3Fr URS
Irreplaceable Clinical Value
1. Significantly Improves Treatment Efficiency
Several papers published in the World Journal of Urology indicate that the 6.3Fr URS demonstrates significantly superior stone-free rates and sheath placement success rates compared to traditional, larger-diameter URS. Using this product can markedly shorten operation time and reduce the incidence of complications.
2. Provides Safer Surgery for Patients with Fragile Ureters
The ureter is long, narrow, and susceptible to damage. Traditional URS often makes a 'no-touch' technique difficult to achieve. For patients with ureteral cancer or coagulation disorders, conventional scopes can potentially cause injury or even severe complications.
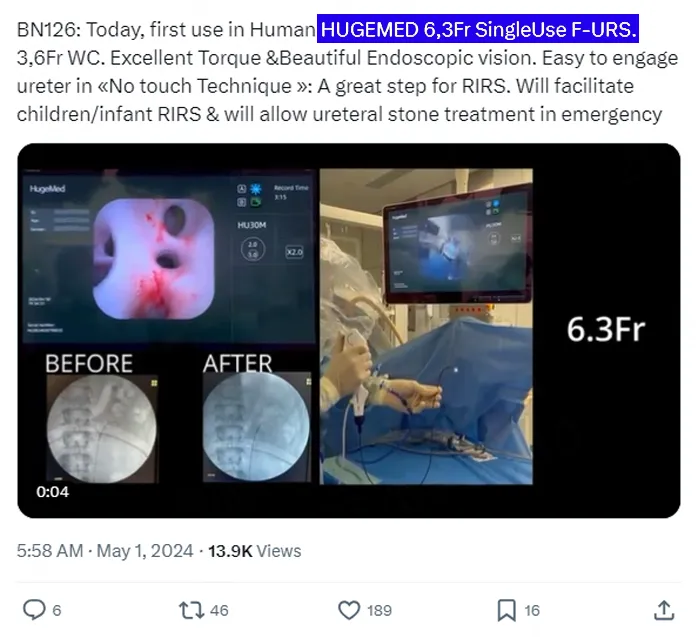
The advent of the 6.3Fr URS offers a safer surgical option for such patients. Renowned French urologist, Professor Olivier Traxer, exclaimed after his first experience: "It makes the 'no-touch technique' much easier to perform. It's a significant step forward for RIRS surgery."
3. Offers a New Surgical Pathway for Patients with Ureteral Strictures or Emergency Obstructions
In cases of congenital or pathological ureteral strictures, traditional URS often cannot pass through smoothly. Physicians typically need to implant a stent and perform dilation before surgery. However, the outer diameter of the 6.3Fr URS is close to that of a standard double-J stent, allowing direct surgery in some emergency stone impaction cases, significantly improving treatment efficiency.
4. Opens Up More Treatment Possibilities for Patients with Complex Anatomy
For patients with complex urinary tract structures such as horseshoe kidneys or duplicated collecting systems, the 6.3Fr URS, with its enhanced reach and flexibility, enables physicians to access and treat areas that were previously unreachable for exploration and treatment.
Multicenter Clinical Studies & Expert Consensus
From "Usable" to "Verifiable"
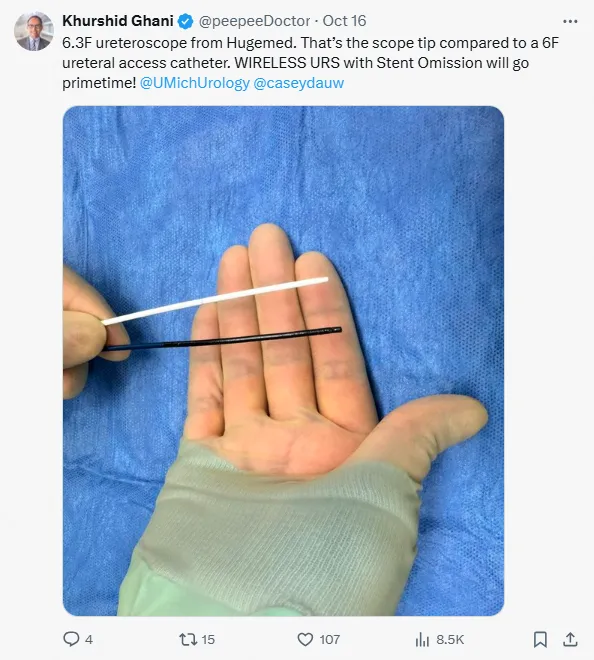
Leveraging these clinical advantages, the 6.3Fr URS has rapidly become a global bestseller since its launch in 2024, setting the industry benchmark. Clinically, HugeMed's 6.3Fr URS has gained recognition worldwide from experts including Professor Li Jianxing, Professor Zeng Guohua, Professor Olivier Traxer (France), Professor Khurshid Ghani (USA), Dr. Mohsen Elmekresh (UAE), and Dr. Vineet Gauhar (Singapore).
Professor Zeng Guohua
Vice President, The First Affiliated Hospital of Guangzhou Medical University
Chairman & Secretary General, International Alliance of Urolithiasis (IAU)
In clinical practice, the value of the 6.3Fr ultra-thin flexible ureteroscope is:
For routine urinary stone patients, the 6.3Fr flexible ureteroscope, with its smaller scope-to-sheath ratio, effectively reduces intrarenal pressure during surgery, thereby lowering the risk of postoperative infectious complications.
When managing special cases such as ureteral strictures, pediatric stones, or patients with coagulation disorders, this scope allows for the use of smaller FANS sheaths, significantly reducing the risk of ureteral injury and bleeding, and avoiding the need for secondary surgeries. Particularly for renal calyceal stones with a narrow infundibulum, the 6.3Fr flexible ureteroscope can directly enter the target area for lithotripsy without needing to incise or dilate the infundibulum, maximizing tissue preservation while ensuring effective treatment.。
In emergency stone removal or tubeless RIRS scenarios, its slim design supports immediate procedural access. In suitable indications, it can eliminate the step of pre-stenting, shortening the overall treatment cycle and reducing the use of associated consumables.
Professor Li Jianxing
Deputy Head, Department of Surgery & Director, Department of Urology, Beijing Tsinghua Changgung Hospital
Vice President & Secretary-General, Chinese Urological Doctor Association (CUDA)
“For general stone patients, the 6.3Fr ultra-thin outer diameter provides more space for stone fragment passage and fluid drainage. This not only improves the single-session stone-free rate but also makes intrarenal pressure and temperature more controllable during surgery, significantly reducing the risk of postoperative edema and infection compared to procedures using previous 7.5Fr+ flexible ureteroscopes.
For high-risk patients, such as those with ureteral strictures, tumors, or coagulation disorders, the 6.3Fr ultra-thin flexible ureteroscope can be used with a 'bare scope' sheathless technique. This can greatly reduce the probability of ureteral injury and intraoperative bleeding.
Due to the slimmer profile and smaller deflection radius of the 6.3Fr scope, it offers more possibilities for difficult ureters and anatomical anomalies like horseshoe kidneys, duplicated systems, and patients with narrow or unfavorable calyceal infundibula. The performance of this product in these challenging situations is currently irreplaceable by other similar products on the market.
Professor Olivier Traxer
Head of Urology Department, Tenon Hospital, Sorbonne University, Paris
President, World Congress of Endourology (WCE) 2018, Paris
"(This 6.3Fr flexible ureteroscope) makes the 'no-touch technique' easier to perform in the ureter. It is a great step forward for RIRS technology."
“Easy to engage ureter in 《 No touch Technique 》: A great step for RlRS.”
On June 7, 2025, HugeMed, in collaboration with Beijing Tsinghua Changgung Hospital, initiated a multicenter, randomized controlled trial: *"A Multicenter Randomized Controlled Trial on the Efficacy and Safety of the 6.3Fr Single-Use Flexible Ureteroscope (Sheathless Technique) versus the 7.5Fr Single-Use Flexible Ureteroscope (Sheath Required) for Treating Upper Urinary Tract Stones < 1.5cm in Diameter."* This study, involving 21 top-tier hospitals, aims to build a robust clinical data system for the 6.3Fr URS in China, focusing on endpoints such as:
Stone-free rate at 1-month post-op
Primary success rate
Operation time
Intraoperative injury rate
Postoperative ureteral stricture rate
Perioperative complications
Postoperative hospital stay (days)
Total cost
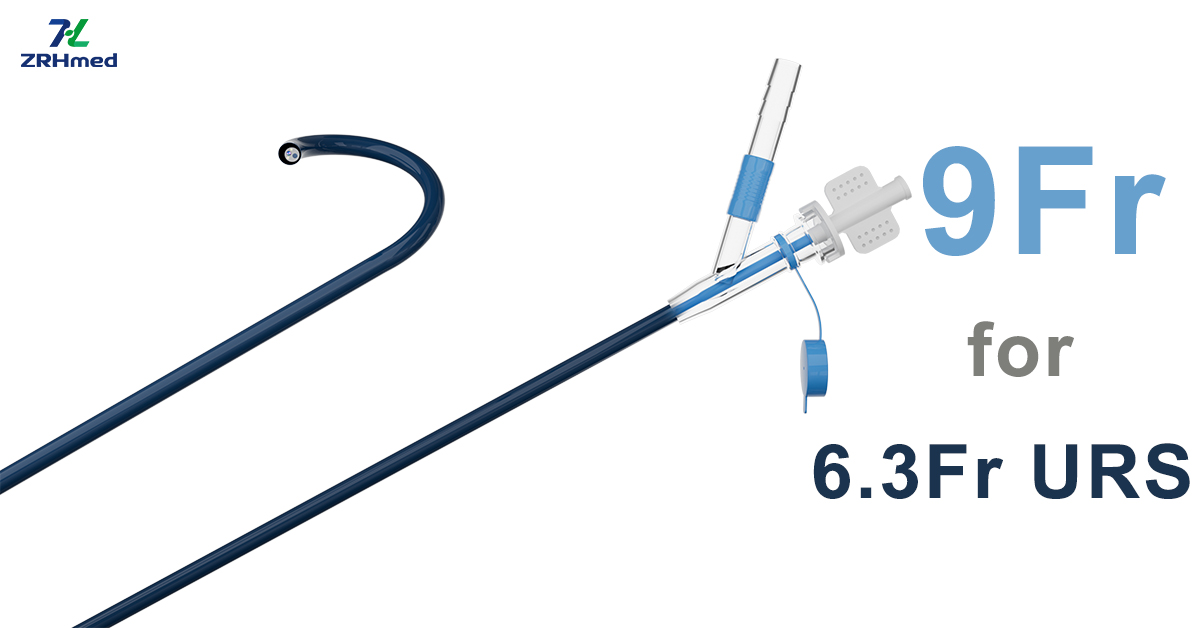
ZRHmed Exclusively Supplies 9Fr UAS with Suction Compatible with 6.3Fr Single-use Ureterorenoscope
To complement your 6.3FR ureteroscope, ZRHmed is the exclusive provider of a specialized 9FR suction sheath engineered for optimal compatibility and performance. This sheath ensures reliable suction, enhanced visibility, and smoother instrument navigation during ureteroscopic procedures. Designed to improve fluid management and stone fragment clearance, it serves as an essential accessory for efficient urological interventions. Trust ZRHmed for exclusive, high-quality solutions tailored to meet the demands of modern endoscopy.
We, Jiangxi Zhuoruihua Medical Instrument Co.,Ltd., is a manufacturer in China specializing in the endoscopic consumables, include GI line such as biopsy forceps, hemoclip, polyp snare, sclerotherapy needle, spray catheter, cytology brushes, guidewire, stone retrieval basket, nasal biliary drainage catheter etc. which are widely used in EMR, ESD, ERCP. And Endourology Line, such as ureteral access sheath with suction, ureteral access sheath, disposable Urinary Stone Retrieval Basket, and urology guidewire etc.
Our products are CE certified, and our plants are ISO certified. Our goods have been exported to Europe, North America, Middle East and part of Asia, and widely obtains the customer of the recognition and praise!




Post time: Mar-04-2026