This exhibition, centered around endoscopy, provided a different feeling and reflection.
Changes in Visitor Traffic
Based on past experience, the visitor flow was expected to be extremely dense, with long queues for both entry and exit, and crowded subways. This time, however, it was less crowded than expected—perhaps due to it being a Thursday. Entering and exiting the venue was notably smooth and fast.
Changes in Visitor Demographics
A second noticeable change was the significant increase in international visitors—Caucasian, Asian, and Black—truly living up to the “International” in “China International Medical Equipment Fair.” International visitors were seen stopping by robotic systems, endoscope lenses, complete systems, portable scopes, and even engaging in discussions about endoscopic carts. On every aisle, foreign attendees could be easily spotted, and many booths had small groups of international visitors engaged in business discussions. It struck me suddenly: the medical endoscopy sector has become a two-way exchange, and increasingly, an outward flow of technology and products.
Advancements in Endoscope Manufacturing
Among pure-play endoscope manufacturers, there were no particularly large booths this time. Some companies even shared exhibition space, reflecting a more pragmatic and grounded approach.
Rigid scopes have become standardized, with 4K or 4K fluorescence as the default. Thanks to the widespread adoption of 4K imaging solutions, image quality has improved significantly. The days of dark, grainy images are gone.
Numerous flexible endoscopes were on display. The handling feel of flexible scopes ranged from very good to at least acceptable—new gastrointestinal (GI) scopes, for example, showed no stiffness or roughness in operation.
However, there are still some minor details that may need refinement:
Color saturation: Colors can be overly saturated and heavy, which may increase operator fatigue over long periods. This is, of course, subject to personal preference.
Design lacks real-world application experience: For instance, some designs make it difficult to hang water bottles properly, creating a severe height mismatch. Such issues could be easily solved by placing the hook on the main unit or on the side of the cart.
Gaps in soft rubber sheath: On a few products, noticeable gaps were observed at the junction between the handle’s lower end and the scope’s shaft—significantly larger than those seen on leading GI scope brands.
Missing on-screen information: Some GI scopes adopt the same centered-image design as rigid scopes, failing to display on-screen endoscope information such as model number, working channel diameter, or shortcut key functions, which may cause minor inconvenience to users.
Ergonomics: Index finger shortcuts were too flat, requiring more force to press. Some handles were heavy or too thick in grip.
The above observations are merely details noted in a critical, almost nitpicking spirit. Wishing the endoscope manufacturing industry continued success in reaching new heights.
Industry Exchange and Conversations
With a constant flow of people—rigid, flexible, and single-use scopes—the endoscopy industry has moved from being niche to ubiquitous, with growth far exceeding a simple “explosion.” So many companies now produce endoscopes, and even more are planning to enter the field.
However, profit margins from manufacturing to end-user have become incredibly transparent and severely compressed. This is why endoscope manufacturers are starting to focus on customer acquisition costs and are eager to export their products overseas.
Some say: “Market share is almost entirely captured by the top few players. How will the dozens or hundreds of smaller companies survive? Maybe being acquired is a good option.”
Some say: “We’ve survived this long because our owner has other businesses. If we were only in endoscopy, we might have gone under. Even when going abroad, it’s still Chinese companies competing fiercely against each other.”
Others note: “Many of the scopes shown here are OEM-produced by their company.” Some ask: “What’s the capacity for single-use cystoscopes? When will single-use GI scopes take off? Will the company survive until then?”
Others share: “Our company started by servicing foreign rigid scopes, then moved to manufacturing our own rigid scopes, and later developed our own endoscope testing systems—from first-generation manual to second-generation automatic. We’re both the athlete and the referee.”
 Despite industry margin compression and some uncertainty, there is still passion for their brands and the endoscopy field. Companies are actively planning how to export more, optimize products, manage teams to improve professionalism, and build stronger organizations. Smiles were seen on most faces.
Despite industry margin compression and some uncertainty, there is still passion for their brands and the endoscopy field. Companies are actively planning how to export more, optimize products, manage teams to improve professionalism, and build stronger organizations. Smiles were seen on most faces.
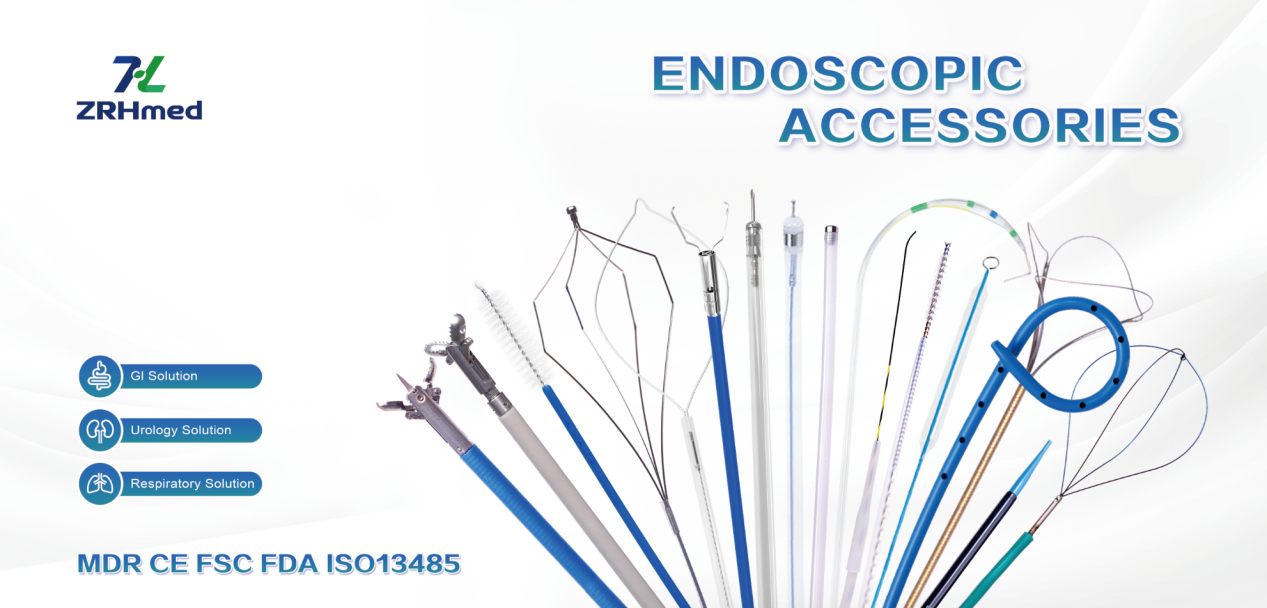
Pro Tip: While large booths are impressive, don’t forget to check the accessory suppliers. Reliable consumables are just as critical for your daily procedures as the scopes themselves.
A quick word from ZRHmed: We specialize in endoscopic accessories, Jiangxi Zhuoruihua Medical Instrument Co.,Ltd., is a manufacturer in China specializing in the endoscopic consumables, include GI line such as biopsy forceps, hemoclip, polyp snare, sclerotherapy needle, spray catheter, cytology brushes, guidewire, stone retrieval basket, nasal biliary drainage cathete etc. which are widely used in EMR, ESD, ERCP. And Endourology Line, such as ureteral access sheath and ureteral access sheath with suction, disposable Urinary Stone Retrieval Basket, and urology guidewire etc.
Our products are CE certified and with FDA 510K approval, and our plants are ISO certified. Our goods have been exported to Europe, North America, Middle East and part of Asia, and widely obtains the customer of the recognition and praise!
Post time: Apr-10-2026