Content sourced from CMEF (China International Medical Equipment Fair), curated for the endoscopy community. Event date: April 9-12, 2026, Venue: National Exhibition and Convention Center (NECC), Shanghai
The Ultimate Guide
|
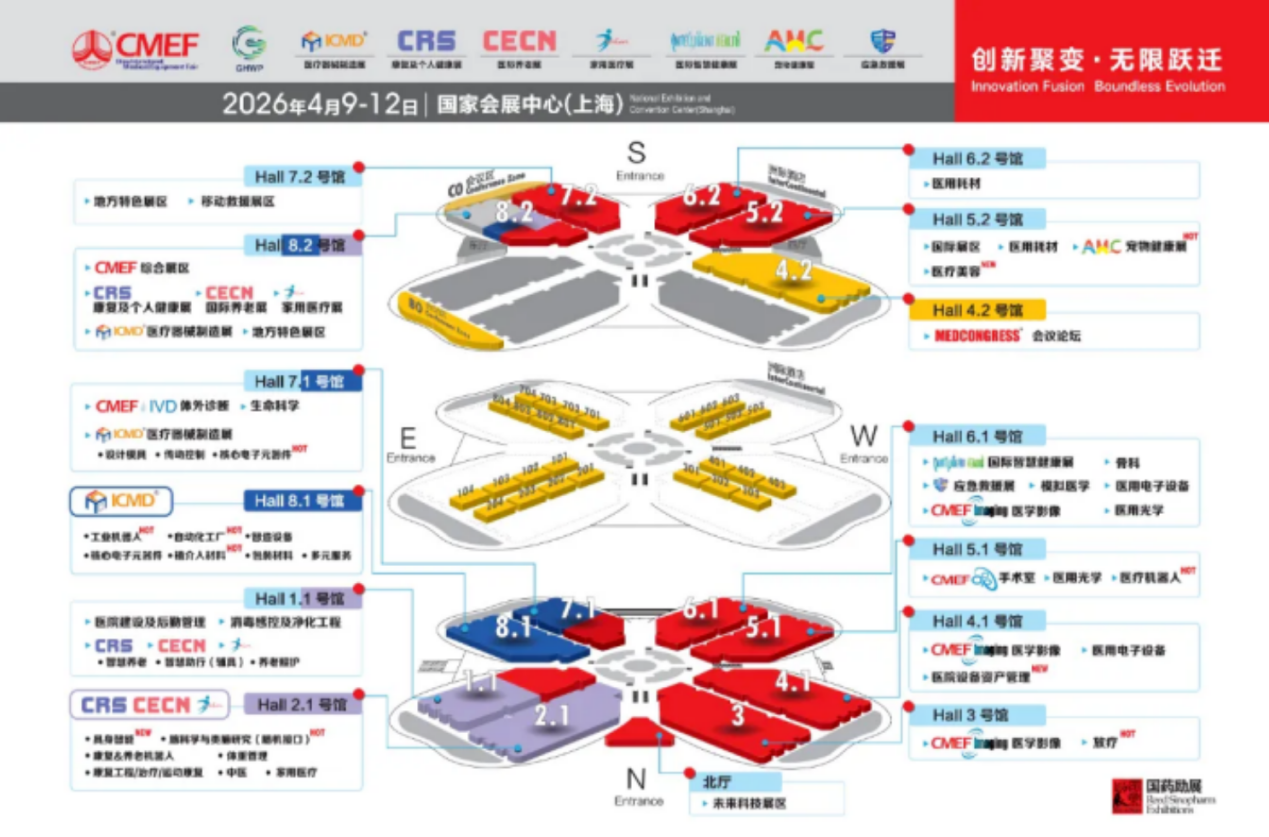
Hall 1.1 |
Hospital Construction & Facility Management, Sterilization & Infection Control, and Purification Engineering |
|
Hall 2.1 |
Embodied Intelligence (NEW), Brain Science and Brain-like Research (Brain-Computer Interface), Rehabilitation & Elderly Care Robots, Weight Management, Rehabilitation Engineering/Therapy/Sports Rehabilitation, Traditional Chinese Medicine, Home Healthcare |
|
Hall 3 |
Medical Imaging & Radiotherapy |
|
Hall 4.1 |
Medical imaging, medical electronic equipment, hospital equipment asset management |
|
Hall 4.2 |
ICMD (Medical Device Manufacturing Exhibition), Regional Characteristic Exhibition Zone |
|
Hall 5.1 |
Operating Room, Medical Optics, Medical Robots |
|
Hall 5.2 |
International Exhibition Area, Medical Consumables, Pet Health Exhibition, Medical Aesthetics, CRS (Rehabilitation and Personal Health Exhibition), CECN (Home Medical Exhibition) |
|
Hall 6.1 |
International Smart Health Exhibition, Orthopedics, Emergency Rescue Exhibition, Simulation Medicine, Medical Electronic Equipment, Medical Imaging, Medical Optics |
|
Hall 6.2 |
Medical consumables |
|
Hall 7.1 |
CMEF IVD (In Vitro Diagnostics), Life Sciences, ICMD Medical Device Manufacturing Exhibition |
|
Hall 7.2 |
Local Specialty Exhibition Area, Mobile Rescue Exhibition Area |
|
Hall 8.1 |
Medical imaging, medical optics, industrial robots, automated factories, intelligent manufacturing equipment, core electronic components, implantable materials, packaging materials |
|
Hall 8.2 |
CMEF includes a comprehensive exhibition area, a local specialty exhibition area, a rehabilitation and personal health exhibition, an international senior care exhibition, and a home medical exhibition. |
|
North Hall |
Future Technology Exhibition Area |
The “Golden Zone” for Endoscopy: Hall 5.1
(Covering Medical Optics, Operating Rooms, & Robotics)
Hall 5.1 is one of the most densely populated venues, hosting approximately 271 exhibitors.
Important Note: It has been confirmed that major endoscopy brands such as Olympus, Karl Storz, Sonoscape, and Aohua will not be participating in this exhibition.
What’s inside?
Beyond the main equipment, Hall 5.1 is also home to endoscopic consumables and upstream supply chain companies, such as Xinze Yuan (Booth 5.1J55) and Suoying (Booth 5.1ZA47-1).
Pro Tip:
If your time and energy allow, we highly recommend visiting Halls 3, 4.1, and 6.1 as well. Many companies have diversified product lines (not just endoscopy) and are therefore located in other halls.
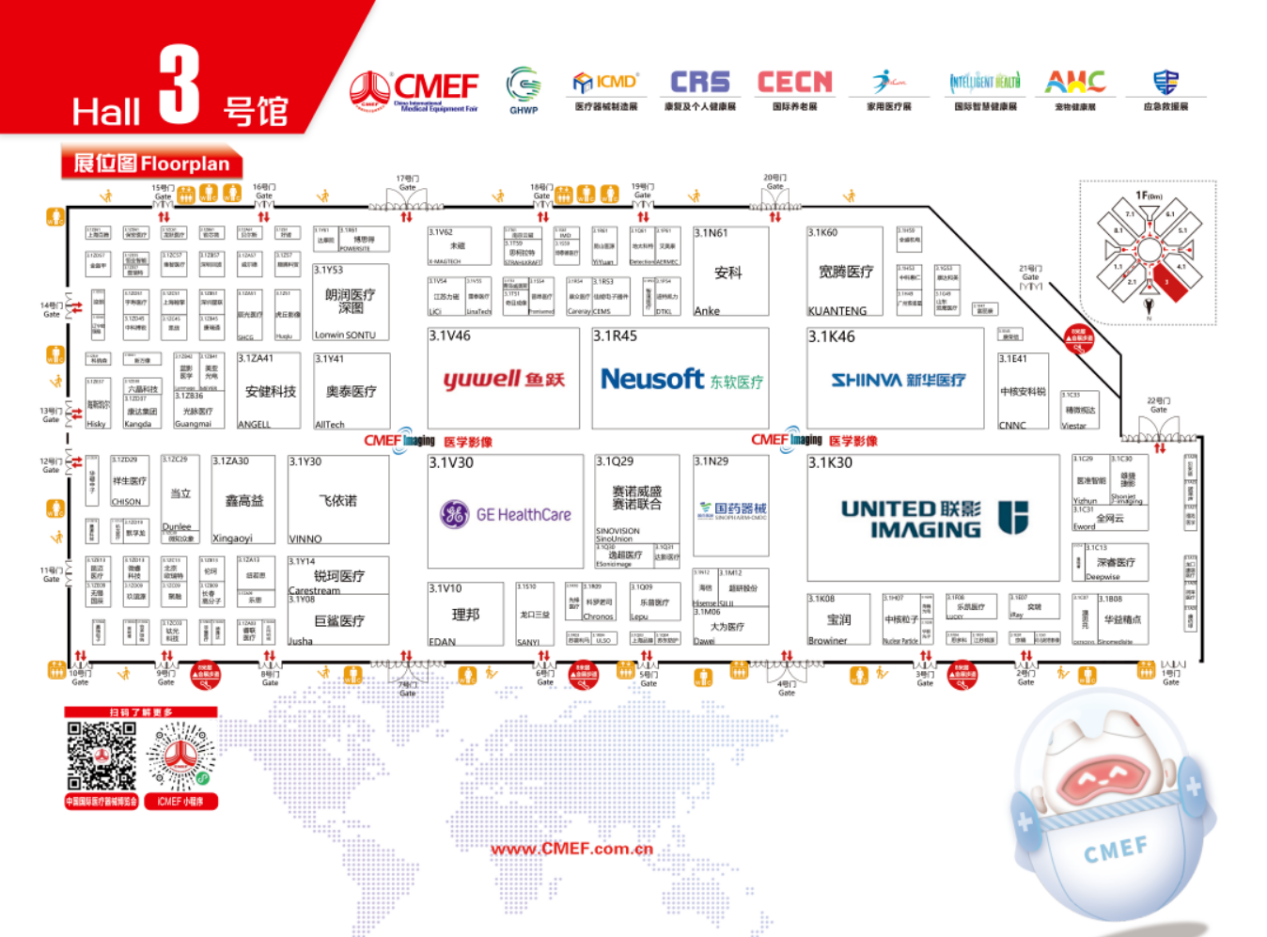
Hall 3: Endoscope Brands & Highlights
Don’t miss these exhibitors in Hall 3:
Edan : Booth 3.1V10
United Imaging: Booth 3.1K30
Lepu Medical: Booth 3.1Q09
VINNO: Booth 3.1Y30
BIOPSEE: Booth 3.1C33
Xinhua: Booth 3.1K46
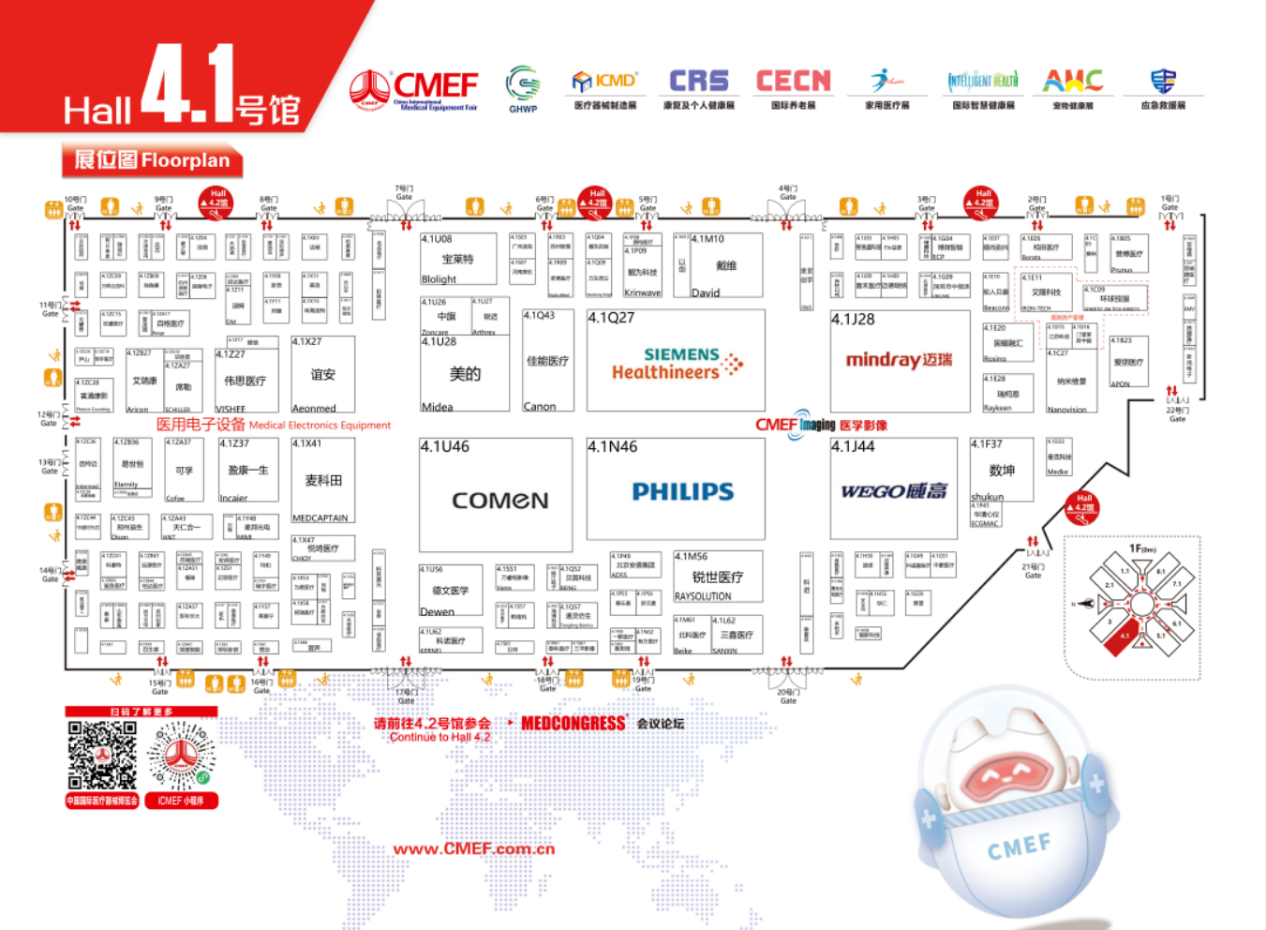
Hall 4.1: Endoscope Brands & Highlights
Check out these key exhibitors in Hall 4.1:
Mindray: Booth 4.1J28
Wego: Booth 4.1J44
Medcaptain: Booth 4.1X41
Comen: Booth 4.1U46
Shanghai Jingying (Software): Booth 4.1H58
Shenzhen ITOOKER: Booth 4.1Q61
Hall 6.1: Endoscopy & Orthopedics
Here are the key brands to look for in Hall 6.1:
Stryker : Booth 6.1T61
Chunli (Orthopedics): Booth 6.1Q38
Tongjin: Booth 6.1ZA53
Hidden Gems in Other Halls
Don’t miss these brands located in other venues:
Ambu (Disposable Endoscopy): Booth 6.2X52
Happiness Works (Disposable Endoscopy): Booth 5.2N59
Gongjiang (Reusable Endoscopy): Booth 5.2K47
Highlighs
- Shanghai Reunion: Industry Networking
- Endoscopy & Robotics: Looking for flexible robotic endoscopes—focusing on both technology and business opportunities. There is also a corresponding forum on flexible robotics in Meeting Room A1, Hall 4.2, on the morning of April 9th.
- Endoscopy & AI: The significance of AI integration for hospital development; understanding national strategies; exploring the current status of intelligent diagnosis, screening, and surgical systems.
- Endoscopy Selection: In this booming field, compare manufacturers across 4K, Full HD, 5mm, and 3mm flexible scopes to experience the highest product standards. It is recommended to use the same reference object for comparison; the palm of the hand is the most convenient reference.
-
Resolution: Differences between 8K, 5K, and 4K; degree of homogeneity vs. differentiated products.
- Image Quality: Noise, handling of over/underexposure, detail display.
- Ease of Operation: Grip feel (rigid & flexible scopes); test deflection—flick quickly to check smoothness, turn slowly to check stability.
- Viewing Comfort: Color tone preference, depth perception, sensation of single-color screen saturation.
5. Minimally Invasive Insights: Discussing the direction and pace of future minimally invasive development with manufacturers, and their advance layout of products and accessories.
6.Supply Chain: Looking at the industrial supply behind endoscopes, from design to production, and from solutions to components, to optimize existing enterprise endoscopes.
7. Product Layout: Observing different manufacturers’ layouts for endoscopy—compatibility, multi-scope vs. single-scope, and whether there are other supporting equipment or consumables besides endoscopes.
Pro Tip: While large booths are impressive, don’t forget to check the accessory suppliers. Reliable consumables are just as critical for your daily procedures as the scopes themselves.
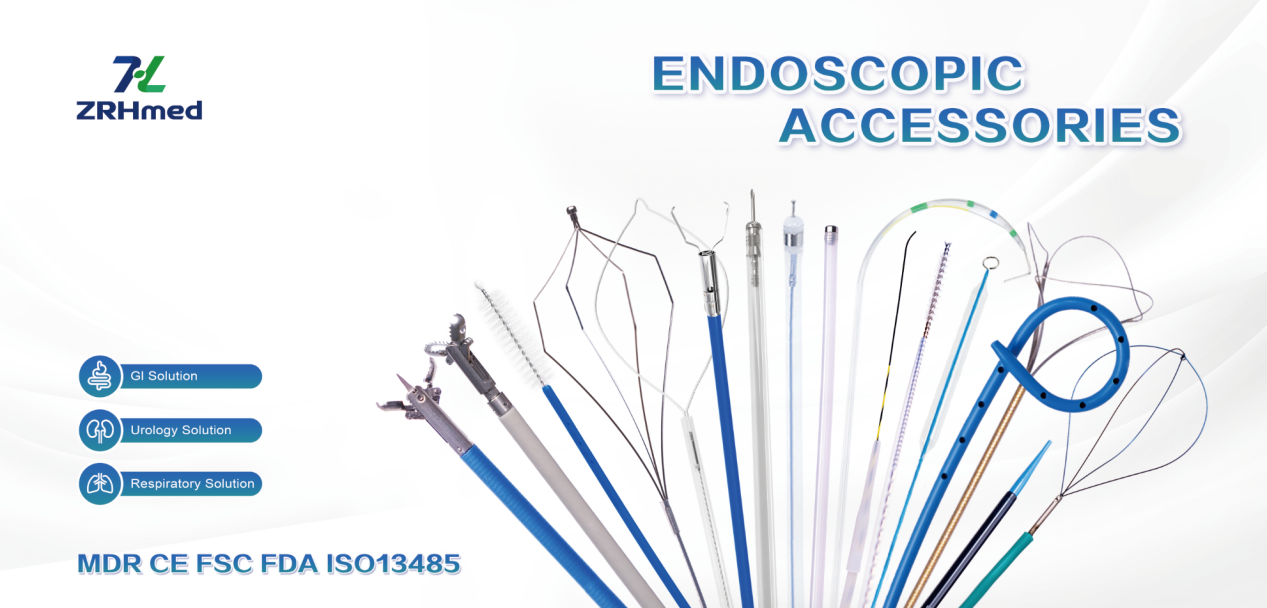
A quick note from us (ZRHmed):We, Jiangxi Zhuoruihua Medical Instrument Co.,Ltd., is a manufacturer in China specializing in the endoscopic consumables, include GI line such as biopsy forceps, hemoclip, polyp snare, sclerotherapy needle, spray catheter, cytology brushes, guidewire, stone retrieval basket, nasal biliary drainage cathete etc. which are widely used in EMR, ESD, ERCP. And Endourology Line, such as ureteral access sheath and ureteral access sheath with suction, disposable Urinary Stone Retrieval Basket, and urology guidewire etc.
Our products are CE certified and with FDA 510K approval, and our plants are ISO certified. Our goods have been exported to Europe, North America, Middle East and part of Asia, and widely obtains the customer of the recognition and praise!
Post time: Apr-08-2026